Anionic surfactants contain negatively charged functional groups at their head:
These functional groups can be sulfate, sulfonate, phosphate or carboxylates. Common alkyl sulfates are ammonium lauryl sulfate, sodium lauryl sulfate (sodium dodecyl sulfate, SLS, or SDS), and the related alkyl-ether sulfates sodium laureth sulfate (sodium lauryl ether sulfate or SLES), and sodium myreth sulfate.
The oldest example of an anionic surfactant is common soap, or sodium stearate:
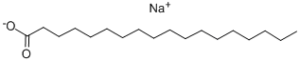
In wax emulsions the most used anionic surfactants are the salts of Oleic acid. Often the counter ions include alkali metal ions like sodium and potassium, or volatile amines or ammonia in case hydrophobicity is needed.